
Stress can have harmful health effects on the body, including the gut microbiome. A recent article in the medical site Medscape reviewed studies of chronic stress and the gut microbiome and found that chronic stress alters the gut microbiome, resulting in gut dysbiosis (the gut microbial community is imbalanced or out-of-whack).
For example, researchers have found alterations in gut microbes, less diversity of microbes, and fewer numbers of certain beneficial microbes in those with PTSD (post-traumatic stress disorder).
Interestingly, the researchers stress that a good diet (e.g., Mediterranean diet, including fermented foods) will help the gut microbiome, but that there isn't any evidence for probiotic supplements. In other words, don't take store-bought daily supplements, but instead focus on eating well, exercise, and getting enough sleep.
From Medscape: How Chronic Stress Disrupts the Gut Microbiome
Chronic psychological stress is common. A 2023 survey revealed that about one quarter of US adults reported high stress levels, and three quarters reported that chronic stress affects their daily lives.
Emerging evidence suggests that chronic stress not only exacts a high toll on mental health but also can wreak havoc on all levels of gastrointestinal (GI) functioning, all the way down to the microbiome. ...continue reading "Chronic Stress and the Gut Microbiome"


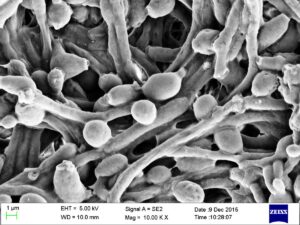
 It's good to eat and drink dairy foods! Consuming dairy foods is beneficial for the gut microbiome, but each dairy food - whether milk, cheese, or yogurt - appears to have different effects on the composition of the gut microbiome (the community of bacteria, viruses, fungi).
It's good to eat and drink dairy foods! Consuming dairy foods is beneficial for the gut microbiome, but each dairy food - whether milk, cheese, or yogurt - appears to have different effects on the composition of the gut microbiome (the community of bacteria, viruses, fungi).

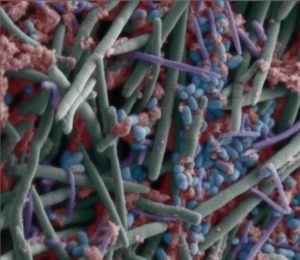

 And what is a health-promoting diet?
And what is a health-promoting diet?
 Recent research found that flare-ups of the disease lupus involves microbial changes in the gut microbiome. The gut microbiome is the community of microbes (fungi, bacteria, viruses) living in the small intestine.
Recent research found that flare-ups of the disease lupus involves microbial changes in the gut microbiome. The gut microbiome is the community of microbes (fungi, bacteria, viruses) living in the small intestine.