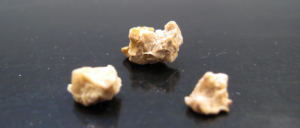
The incidence of kidney stones is on the rise worldwide, with 1 in 11 people expected to develop a stone at some point. There are several subtypes of kidney stones, with calcium oxalate (CaOx) stones occurring in more than 70% of cases. Recent research found live bacteria in calcium oxalate stones, along with biofilms.
Biofilms are communities of bacteria sticking to one another and coated with a protective slime. In these kidney stones, the biofilms were in-between mineral layers. Several bacterial species were most common in the kidney stones: E. faecalis, P. mirabilis, and E. coli.
Prior to this study, calcium oxalate kidney stones were not considered bacterial. The researchers thought that the bacteria might explain why people tend to have recurrent kidney stones.
Kidney stones begin as tiny crystals that can accumulate and clump together in urine. One easy way to decrease risk of developing kidney stones is do drink plenty of water (want to dilute your urine). Low fluid intake and dehydration is associated with increased kidney stone formation, due to concentrated urine.
An interesting earlier study looked at the microbiome of the kidneys and found that the beneficial bacteria L. crispatus is found in the absence of kidney stones, while the presence of E.coli was associated with the development of kidney stones. The researchers found that the L. crispatus somehow blocked E.coli's ability to form kidney stones.
From Medical Xpress: Previously unknown bacterial component in kidney stone formation discovered
In an unexpected finding, a UCLA-led team has discovered that bacteria are present inside the most common type of kidney stone, revealing a previously unrecognized component involved in their formation. ...continue reading "Bacteria Found In Kidney Stones"

 The microbes in your mouth may offer a clue to obesity. New
The microbes in your mouth may offer a clue to obesity. New 

 There is another easy (and lazy) way to use L. sakei (e.g., Lanto Sinus), for sinusitis or when sinus symptoms are starting. Only when needed, of course.
There is another easy (and lazy) way to use L. sakei (e.g., Lanto Sinus), for sinusitis or when sinus symptoms are starting. Only when needed, of course.
 It is still unknown whether probiotic supplementation would help these conditions because the studies are not yet done. But researchers do suggest eating an anti-inflammatory diet - which means a diet rich in fruits, vegetables, whole grains, legumes (includes beans), fish, seeds, nuts, and olive oil. This kind of diet has lots of fiber to feed and support beneficial bacteria.
It is still unknown whether probiotic supplementation would help these conditions because the studies are not yet done. But researchers do suggest eating an anti-inflammatory diet - which means a diet rich in fruits, vegetables, whole grains, legumes (includes beans), fish, seeds, nuts, and olive oil. This kind of diet has lots of fiber to feed and support beneficial bacteria. Millions of pounds of pesticides are used each year in the US: on farms, in buildings and homes, on lawns, in pet flea and tick products - basically everywhere. And so the pesticides eventually wind up in us - from the foods we eat, the water we drink, the air we breathe (when pesticides drift during applications or when used indoors), and the treated lawns and ground we walk on.
Millions of pounds of pesticides are used each year in the US: on farms, in buildings and homes, on lawns, in pet flea and tick products - basically everywhere. And so the pesticides eventually wind up in us - from the foods we eat, the water we drink, the air we breathe (when pesticides drift during applications or when used indoors), and the treated lawns and ground we walk on. Exercise helps fight cancer. Wow! What a headline. But is it true?
Exercise helps fight cancer. Wow! What a headline. But is it true?
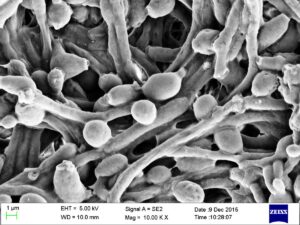
 What foods to avoid or to eat has long been debated in chronic kidney disease treatment. Traditionally, people with chronic kidney disease (CKD) have been advised to avoid a number of fruits and vegetables. In contrast, a recent
What foods to avoid or to eat has long been debated in chronic kidney disease treatment. Traditionally, people with chronic kidney disease (CKD) have been advised to avoid a number of fruits and vegetables. In contrast, a recent