 Last week a person told an amazing story in the comments section after a post on this site. After suffering from a "constant runny nose and a bad smell" in the nose for 2 years - which was diagnosed as "fungi and staph" in the sinuses - the person started doing "kimchi treatments" (as discussed in the Sinusitis Treatment Summary page). After 2 weeks a fungal ball was loosened, which came out of the sinuses and into the mouth, and was then spit out. About an inch in size - a smelly, grey/green, round fungal ball. Wow. Which leads to the question: Are any of the microbes in live kimchi anti-fungal?
Last week a person told an amazing story in the comments section after a post on this site. After suffering from a "constant runny nose and a bad smell" in the nose for 2 years - which was diagnosed as "fungi and staph" in the sinuses - the person started doing "kimchi treatments" (as discussed in the Sinusitis Treatment Summary page). After 2 weeks a fungal ball was loosened, which came out of the sinuses and into the mouth, and was then spit out. About an inch in size - a smelly, grey/green, round fungal ball. Wow. Which leads to the question: Are any of the microbes in live kimchi anti-fungal?
Kimchi is an amazing live fermented food, typically made with cabbage and other vegetables and a variety of seasonings. Kimchi is the national dish of Korea and so there is tremendous interest in Korea in studying kimchi to learn about the many different microbial species in kimchi, including how they change over the course of fermentation.
It turns out that kimchi contains many species of bacteria, including various species of Lactobacillus - which are considered beneficial. Of course one of the species found in kimchi over the course of fermentation is Lactobacillus sakei - the bacteria that successfully treats sinusitis, and which I have written about extensively. L. sakei predominates over pathogenic bacteria (antibacterial) - which is why it is also used as a sausage starter culture (to kill off bacteria such as Listeria). One study found that the garlic, ginger, and leek used in making kimchi were the sources of L. sakei bacteria found in fermented kimchi.
Studies show that a number of the Lactobacillus species found in kimchi are antifungal against a number of different kinds of fungi. Some of these antifungal bacteria are: Lactobacillus plantarum, L. cruvatus, L. lactis, L. casei, L. pentosus, L. acidophilus, and L. sakei (here, here).
A study from 2005 found that some Lactobacillus species found in kimchi are predominant over a fungi known to cause health problems in humans - Aspergillus fumigatus, a mold (fungi) which is the most common cause of Aspergillus infections. Aspergillus (of which there are many species) is very common both indoors and outdoors (on plants, soil, rotting plants, household dust, etc.), so people typically breathe in these fungal spores daily and without any negative effects. However, sometimes Aspergillus can cause allergic reactions, infections in the lungs and sinuses (including fungal balls), and other infections. (more information at CDC site).
The study found that 5 bacterial species in kimchi were also antifungal against other species of fungi (Aspergillus flavus, Fusarium moniliforme, Penicillium commune, and Rhizopus oryzae). The 5 bacterial species in kimchi that they found to be antifungal were: Lactobacillus cruvatus, L. lactis subsp. lactis, L. casei, L. pentosus, and L. sakei.
Just keep in mind that fungi are everywhere around us, and even part of the microbes that live in and on us - this is our mycobiome. We also breathe in a variety of fungi (mold spores) every day. In healthy individuals (even babies) all the microbes (bacteria, viruses, fungi, etc) live in balanced microbial communities, but the communities can become "out of whack" (dysbiosis) for various reasons, and microbes that formerly co-existed peacefully can multiply and become problematic.
If the populations get too unbalanced (e.g., antibiotics can kill off bacteria, and then an increase in fungi populations take their place) then ordinarily non-harmful fungi can become pathogenic. Or other pathogenic microbes can enter the community (e.g., through infection), and the person becomes ill.
IN SUMMARY: Kimchi has beneficial bacteria in it that are effective not just against bacteria (antibacterial), but also against some kinds of fungi (antifungal). One 2016 review study went so far as to say: "Kimchi possesses anti-inflammatory, antibacterial, antioxidant, anticancer, antiobesity, probiotic properties, cholesterol reduction, and antiaging properties."
Experiences of my family and people writing suggest that the L. sakei in kimchi (and other products) is also antibiofilm. Hopefully, there will be some research on this in the future. But in the meantime, please keep writing to me about fungal complications of sinusitis, and especially if kimchi, L. sakei products, or other probiotics helped.

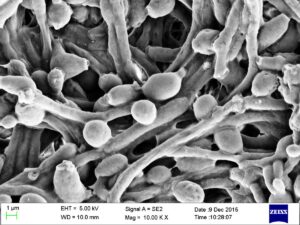

 The sinus microbiome consists of bacteria, viruses, and fungi all coexisting in the sinuses. Research has long focused on how the bacteria in sinus microbiomes in people with chronic sinusitis is different than in healthy persons without sinusitis (and yes, there are differences). Differences in the mycobiome (the fungi) of the
The sinus microbiome consists of bacteria, viruses, and fungi all coexisting in the sinuses. Research has long focused on how the bacteria in sinus microbiomes in people with chronic sinusitis is different than in healthy persons without sinusitis (and yes, there are differences). Differences in the mycobiome (the fungi) of the 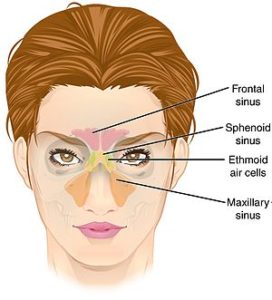
 We all know that microbes (fungi, viruses, bacteria) live throughout our bodies - this is the human microbiome or microbiota. What is really interesting is that cancer tumors also have microbiomes (tumor microbiome), and these microbial communities are different than that found in healthy people (without tumors).
We all know that microbes (fungi, viruses, bacteria) live throughout our bodies - this is the human microbiome or microbiota. What is really interesting is that cancer tumors also have microbiomes (tumor microbiome), and these microbial communities are different than that found in healthy people (without tumors). Many studies show that antibiotics disrupt the gut microbiome (intestinal microbial community of bacteria, viruses, fungi) in adults, but what about infants? A
Many studies show that antibiotics disrupt the gut microbiome (intestinal microbial community of bacteria, viruses, fungi) in adults, but what about infants? A 
 Researchers have known for a while that human breast milk contains hundreds of
Researchers have known for a while that human breast milk contains hundreds of  How many people know this? That wallpaper could have fungi (mold) living on it, and this fungi can release toxins (mycotoxins) that can pollute the air and sicken people when people inhale the toxins. The releasing of toxins from the fungi (mold) into the air is called
How many people know this? That wallpaper could have fungi (mold) living on it, and this fungi can release toxins (mycotoxins) that can pollute the air and sicken people when people inhale the toxins. The releasing of toxins from the fungi (mold) into the air is called  Last week a person told an
Last week a person told an  A new study has summarized what we know about fungi that live in and on babies - and yes, we all have fungi both on and within us. It's called the mycobiome. In healthy individuals all the microbes (bacteria, viruses, fungi, etc) live in balanced microbial communities, but the communities can become "out of whack" (dysbiosis) for various reasons, and microbes that formerly co-existed peacefully can multiply and become problematic. Or other pathogenic microbes can enter the community, and the person becomes ill.
A new study has summarized what we know about fungi that live in and on babies - and yes, we all have fungi both on and within us. It's called the mycobiome. In healthy individuals all the microbes (bacteria, viruses, fungi, etc) live in balanced microbial communities, but the communities can become "out of whack" (dysbiosis) for various reasons, and microbes that formerly co-existed peacefully can multiply and become problematic. Or other pathogenic microbes can enter the community, and the person becomes ill. Could this be? Fungal infection being the cause of Alzheimer's disease? Noteworthy from a recent study conducted in Spain: all the Alzheimer's disease (AD) patients had evidence of fungal infections in their brains, central nervous systems, and vascular systems, but none were found in the control subjects (those without Alzheimer's disease). Many of the symptoms of AD (such as inflammation of the central nervous system and activation of the immune system) match those with long-lasting fungal infections. A "microbial cause" has long been suggested as a cause of AD, and interestingly other studies have also found fungal infections in AD patients. The research so far has found
Could this be? Fungal infection being the cause of Alzheimer's disease? Noteworthy from a recent study conducted in Spain: all the Alzheimer's disease (AD) patients had evidence of fungal infections in their brains, central nervous systems, and vascular systems, but none were found in the control subjects (those without Alzheimer's disease). Many of the symptoms of AD (such as inflammation of the central nervous system and activation of the immune system) match those with long-lasting fungal infections. A "microbial cause" has long been suggested as a cause of AD, and interestingly other studies have also found fungal infections in AD patients. The research so far has found