 Try to avoid triclosan. Read labels (especially soaps, personal care, and household cleaning products) and avoid anything that says it contains triclosan, or is anti-bacterial, anti-fungal, anti-microbial, or anti-odor. We easily absorb triclosan into our bodies, and it has been detected in our urine, blood, and breast milk. Among its many negative effects (e.g., here and here) is that it is now linked to disruption of gut bacteria.
Try to avoid triclosan. Read labels (especially soaps, personal care, and household cleaning products) and avoid anything that says it contains triclosan, or is anti-bacterial, anti-fungal, anti-microbial, or anti-odor. We easily absorb triclosan into our bodies, and it has been detected in our urine, blood, and breast milk. Among its many negative effects (e.g., here and here) is that it is now linked to disruption of gut bacteria.
The gut microbiome performs vital functions in our bodies. Deviation from the normal microbiome (our microbial communities being out of whack) is known as dysbiosis. Dysbiosis has been associated with human diseases, including diabetes, heart disease, arthritis, and malnutrition. The researchers used zebrafish because they are commonly used to study impacts of various chemicals on gut microbial communities. So yes, the findings are valid and match what other studies have found. So please try to avoid triclosan. From Science Daily:
Common antimicrobial agent rapidly disrupts gut bacteria
A new study suggests that triclosan, an antimicrobial and antifungal agent found in many consumer products ranging from hand soaps to toys and even toothpaste, can rapidly disrupt bacterial communities found in the gut.The research was published in PLOS ONE by scientists from Oregon State University. It was based on findings made with zebrafish, which researchers believe are an important animal model to help determine possible human biological and health impacts of this antimicrobial compound.
Triclosan was first used as a hospital scrub in the 1970s and now is one of the most common antimicrobial agents in the world, found in shampoos, deodorants, toothpastes, mouth washes, kitchen utensils, cutting boards, toys, bedding, socks and trash bags. It continues to be used in medical settings, and can be easily absorbed through the skin.
"However, there's now a growing awareness of the importance of the bacteria in our gut microbiome for human health, and the overuse of antibiotics that can lead to the rise of 'superbugs.' There are consequences to constantly trying to kill the bacteria in the world around us, aspects we're just beginning to understand."
In the new study, researchers found that triclosan exposure caused rapid changes in both the diversity and composition of the microbiome in the laboratory animals. It's not clear what the implication may be for animal or human health, but scientists believe that compromising of the bacteria in the intestinal tract may contribute to the development or severity of disease. Some bacteria were more susceptible to the impact of triclosan than others, such as the family Enterobacteriaceae; and others were more resilient, such as the genus Pseudomonas.
The gut-associated microbiome performs vital functions for human health, prevents colonization with pathogens, stimulates the development of the immune system, and produces micronutrients needed by the host. Dysfunction of this microbiome has been associated with human disease, including diabetes, heart disease, arthritis and malnutrition, the scientists pointed out in their study.
Triclosan has been a concern in part because it is so widely used, and it's also readily absorbed through the skin and gastrointestinal tracts, showing up in urine, feces and breast milk. It also has been associated with endocrine disruption in fish and rats, may act as a liver tumor promoter, and can alter inflammatory responses.

 In a newly published study looking at how infant gut microbes change over time, once again babies had differences in gut bacteria depending on whether they were delivered vaginally or by Cesarean section.
In a newly published study looking at how infant gut microbes change over time, once again babies had differences in gut bacteria depending on whether they were delivered vaginally or by Cesarean section. Amusing but also scary. The negative effects on the gut microbes of one person consuming an all fast food diet for 10 days occurred very quickly, and his gut microbes did not recover even 2 weeks after the fast food diet ended. Biggest problem seemed to be loss of gut diversity - about 40% of his gut bacterial species. Loss of gut diversity is considered a sign of ill health. Written by Tim Spector, with Tom Spector's assistance, from The Conversation:
Amusing but also scary. The negative effects on the gut microbes of one person consuming an all fast food diet for 10 days occurred very quickly, and his gut microbes did not recover even 2 weeks after the fast food diet ended. Biggest problem seemed to be loss of gut diversity - about 40% of his gut bacterial species. Loss of gut diversity is considered a sign of ill health. Written by Tim Spector, with Tom Spector's assistance, from The Conversation:
 Tom Spector. Credit: Tim Spector
Tom Spector. Credit: Tim Spector There has been a lot of discussion in the last few years of our
There has been a lot of discussion in the last few years of our 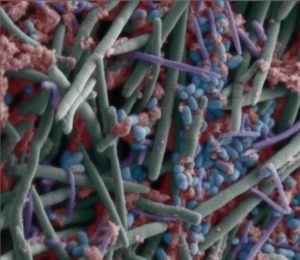 Gut bacteria in children varies among different Asian countries. A
Gut bacteria in children varies among different Asian countries. A