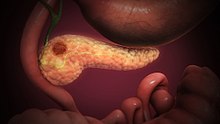
Pancreatic cancer is typically discovered only when it is advanced, very hard to treat, and typically with poor outcomes. Interesting recent research found that the gut microbiome is distinctive in persons with pancreatic cancer, even in the early stages. Perhaps this finding will lead to an easy noninvasive way to screen for pancreatic cancer.
Researchers in Spain and Germany looked at both the community of microbes (microbiome) living in the mouth and the gut (shown in a person's feces). They took samples from healthy persons, individuals with pancreatic cancer, and persons with chronic pancreatitis and found that the bacteria in the stool were predictive for pancreatic cancer (but not those in the mouth).
The "fecal microbiota signature" or pattern of microbes in those with pancreatic cancer had an increase in some microbes (e.g., Fusobacterium nucleatum) and a decrease in others (e.g., Faecalibacterium prausnitzii) - when compared to healthy individuals.
By the way, Fusobacterium nucleatum is found increased in other cancers, such as colon cancer, and beneficial and anti-inflammatory Faecalibacterium prausnitzii is reduced in a number of diseases. Eating a diet rich in high fiber foods (whole grains, fruits, vegetables, legumes, seeds, nuts) increase the numbers of F. prausnitzi in a person's gut microbial community.
From Medical Xpress: Distinct gut microbial profile may identify pancreatic cancer, irrespective of stage
A specific panel of gut microbes may identify pancreatic cancer, irrespective of how far the disease has progressed, suggests research published online in the journal Gut. ...continue reading "Pancreatic Cancer Has a Distinct Gut Microbial Pattern"


 Emulsifiers are in many of the processed foods we buy. They are added to the foods to enhance texture and extend shelf life. Animal and human studies find that some emulsifiers (e.g., soy lecithin, carrageenan, polysorbate-80) can promote gut inflammation and alter the gut microbiome in a
Emulsifiers are in many of the processed foods we buy. They are added to the foods to enhance texture and extend shelf life. Animal and human studies find that some emulsifiers (e.g., soy lecithin, carrageenan, polysorbate-80) can promote gut inflammation and alter the gut microbiome in a  Those who enjoy a little "potty humor" will like the results of a recent
Those who enjoy a little "potty humor" will like the results of a recent  Antibiotics can be life-saving, but there are also unintended consequences. One of them is that they disrupt and alter the gut microbiome (the microbial community of the millions of microbes living in the intestines). A
Antibiotics can be life-saving, but there are also unintended consequences. One of them is that they disrupt and alter the gut microbiome (the microbial community of the millions of microbes living in the intestines). A  Fermented foods include: yogurt, buttermilk, sour cream, cheese, kefir, fermented vegetables, kimchi, natto, miso, sauerkraut, traditional pickles, traditional sourdough bread, apple cider vinegar, and kombucha. To quickly improve the gut microbiome, try to eat six 1/2 cup servings each day for a
Fermented foods include: yogurt, buttermilk, sour cream, cheese, kefir, fermented vegetables, kimchi, natto, miso, sauerkraut, traditional pickles, traditional sourdough bread, apple cider vinegar, and kombucha. To quickly improve the gut microbiome, try to eat six 1/2 cup servings each day for a  There are microbial differences (microbiome) in babies who are born by cesarean vs vaginal deliveries.
There are microbial differences (microbiome) in babies who are born by cesarean vs vaginal deliveries.  The findings of a recent
The findings of a recent  We've known for a while that exposure to microbes during the baby's first year is important (and good) for the developing immune system. In addition, recent research suggests that during the second trimester of pregnancy the developing fetus already has microbes in its body which help "educate" the developing immune system.
We've known for a while that exposure to microbes during the baby's first year is important (and good) for the developing immune system. In addition, recent research suggests that during the second trimester of pregnancy the developing fetus already has microbes in its body which help "educate" the developing immune system. Uh oh... Glyphosate is the most widely used herbicide (plant-killer) in the world, and its pervasive use may be harming our gut microbiomes. Glyphosate (which is in Roundup) is used not only as a weed-killer, but also applied to glyphosate resistant genetically engineered (GE) crops such as soy, canola, corn, and also right before harvest (
Uh oh... Glyphosate is the most widely used herbicide (plant-killer) in the world, and its pervasive use may be harming our gut microbiomes. Glyphosate (which is in Roundup) is used not only as a weed-killer, but also applied to glyphosate resistant genetically engineered (GE) crops such as soy, canola, corn, and also right before harvest (